› Forums › General Melanoma Community › PLX4032 ….please help
- This topic has 28 replies, 11 voices, and was last updated 15 years, 7 months ago by
Jerry from Cape Cod.
- Post
-
- October 13, 2010 at 12:21 pm
I am being offered PLX4032 at Yale. Im not sure if I want to take the drug as I feel like it is a tease. All the research shows that the tumors start dissipating and then after months they come back with a vengeance. Has anyone taking this drug and had long term positive results? What are the side effects? Decisions decisions!! Im so sick of it. Any help would be greatly appreciated.
Lisa
I am being offered PLX4032 at Yale. Im not sure if I want to take the drug as I feel like it is a tease. All the research shows that the tumors start dissipating and then after months they come back with a vengeance. Has anyone taking this drug and had long term positive results? What are the side effects? Decisions decisions!! Im so sick of it. Any help would be greatly appreciated.
Lisa
- Replies
-
-
- October 13, 2010 at 12:43 pm
Lisa,
I am scheduled to start the PLX4032 trial around November 1. I was lucky # 10 out of 10 for my oncologists office. You may need to make your decision quickly before all of the trial slots are taken. After i signed up, my oncologist continued to receive requests to participate in the trial.
I guess the drug could be considered a tease since most, if not all of the trial participants have had their tumors grow after a period of time. I took a look at this option which had a high percentage of patients with a response and compared it to the other options out there. Ipi, which has a 30% response rate but has quite a few respondents with a long term durable response is an option, but only as part of the compassionate use trial.
My thought process was to go on the PLX4032 and hopefully have fantastic tumor reduction results for the maximum period of time, whatever that might be, perhaps 18-24 months or longer. At that time, Ipilimumab would be fully FDA approved and available in more locations and that will probably be my Plan 'B', although I might try one more clinical trial a that time if there are other new and promising treatments available.
This whole process sucks since there is not one treatment that will work for everyone.
Good luck with your decision making process.
Like others on this board have said, Plan a course of action and move forward and hope for the best. Don't second guess yourself and look back and say "if only I had selected a different option". You never know what option will work best for you. You just have to have the strength to keep on trying, although I understand how tough that can be at times.
Bill from Illinois.
-
- October 13, 2010 at 12:43 pm
Lisa,
I am scheduled to start the PLX4032 trial around November 1. I was lucky # 10 out of 10 for my oncologists office. You may need to make your decision quickly before all of the trial slots are taken. After i signed up, my oncologist continued to receive requests to participate in the trial.
I guess the drug could be considered a tease since most, if not all of the trial participants have had their tumors grow after a period of time. I took a look at this option which had a high percentage of patients with a response and compared it to the other options out there. Ipi, which has a 30% response rate but has quite a few respondents with a long term durable response is an option, but only as part of the compassionate use trial.
My thought process was to go on the PLX4032 and hopefully have fantastic tumor reduction results for the maximum period of time, whatever that might be, perhaps 18-24 months or longer. At that time, Ipilimumab would be fully FDA approved and available in more locations and that will probably be my Plan 'B', although I might try one more clinical trial a that time if there are other new and promising treatments available.
This whole process sucks since there is not one treatment that will work for everyone.
Good luck with your decision making process.
Like others on this board have said, Plan a course of action and move forward and hope for the best. Don't second guess yourself and look back and say "if only I had selected a different option". You never know what option will work best for you. You just have to have the strength to keep on trying, although I understand how tough that can be at times.
Bill from Illinois.
-
- October 13, 2010 at 1:45 pm
Thanks Bill. I actually just finished ipi and it still may have time to work but due to the deadline they want me to start BRAF. I dont know!!! I have some real thinking to do today. I know its competitive to get the drug and that everybody wants to get on it but i just dont want a bandaid I want it to go away forever. Best of luck with your course of treatment.
I'll keep you posted on my decision.
Lisa
-
- October 13, 2010 at 1:45 pm
Thanks Bill. I actually just finished ipi and it still may have time to work but due to the deadline they want me to start BRAF. I dont know!!! I have some real thinking to do today. I know its competitive to get the drug and that everybody wants to get on it but i just dont want a bandaid I want it to go away forever. Best of luck with your course of treatment.
I'll keep you posted on my decision.
Lisa
-
- October 13, 2010 at 5:19 pm
There is not any immunosuppression with BRAF as far as I know, the only reason not to do this, in my mind would be it might confuse the response to IPI, which is why I am surprised you are allowed to. Anyway, anything that will reduce tumor burden is a good thing. The only other thing I might consider would be a vaccine trial to boost the effects of IPI.
Best of Luck.
John
-
- October 20, 2010 at 9:50 pm
It's not the AVERAGE it's the Median
The time quoted is not an average it's the mean or median which is quite different. On entering the ipi trial all must have a 16 week MINIMUM life expectancy. I'm not sure of the number but the median life expectancy is 3-4 months beyond that. The average life expectancy of any group will only be known when the last member of that group has died.
With limited access trials many who have to wait for entry might have had a better outcome if the treatment had been available earlier. It's the part of cancer research and drug approval that sucks big time. It is however, better to have the treatment tested and understood before released in mass.
I've been in three trials and take my role seriously and I do everything in my power to understand what the up and downsides are and any other options I may have.
I'm not advocating any treatment over another here, but please get the terminology and the meaning correct so as not to confuse new visitors.
The other issue facing Doctors and Researchers is how to select treatments for the best effect. It's predicted that a time will come when the combination treatments will give the Doctors more effective options for patients along with accurate screening tests.
We all just happen to be in the beginning of the new era of Melanoma treatment
-
- October 20, 2010 at 9:50 pm
It's not the AVERAGE it's the Median
The time quoted is not an average it's the mean or median which is quite different. On entering the ipi trial all must have a 16 week MINIMUM life expectancy. I'm not sure of the number but the median life expectancy is 3-4 months beyond that. The average life expectancy of any group will only be known when the last member of that group has died.
With limited access trials many who have to wait for entry might have had a better outcome if the treatment had been available earlier. It's the part of cancer research and drug approval that sucks big time. It is however, better to have the treatment tested and understood before released in mass.
I've been in three trials and take my role seriously and I do everything in my power to understand what the up and downsides are and any other options I may have.
I'm not advocating any treatment over another here, but please get the terminology and the meaning correct so as not to confuse new visitors.
The other issue facing Doctors and Researchers is how to select treatments for the best effect. It's predicted that a time will come when the combination treatments will give the Doctors more effective options for patients along with accurate screening tests.
We all just happen to be in the beginning of the new era of Melanoma treatment
-
- October 16, 2010 at 1:33 am
When my husband took the IL-2 we were told the response rate was lower than 20% by our oncologist don't remember the exact # though. But we took it anyway because it was the next line of tx.
Linda/Kentucky
-
- October 16, 2010 at 1:33 am
When my husband took the IL-2 we were told the response rate was lower than 20% by our oncologist don't remember the exact # though. But we took it anyway because it was the next line of tx.
Linda/Kentucky
-
- October 13, 2010 at 5:19 pm
There is not any immunosuppression with BRAF as far as I know, the only reason not to do this, in my mind would be it might confuse the response to IPI, which is why I am surprised you are allowed to. Anyway, anything that will reduce tumor burden is a good thing. The only other thing I might consider would be a vaccine trial to boost the effects of IPI.
Best of Luck.
John
-
- October 20, 2010 at 9:28 pm
Have you considered the trial
Dose-escalation Study of Combination BMS-936558 (MDX-1106) and Ipilimumab in Subjects With Unresectable Stage III or Stage IV Malignant MelanomaSeems like a match made in heaven! No placebo!MDX 1106 is showing great promise and is a Anti-PD-1. -
- October 20, 2010 at 9:28 pm
Have you considered the trial
Dose-escalation Study of Combination BMS-936558 (MDX-1106) and Ipilimumab in Subjects With Unresectable Stage III or Stage IV Malignant MelanomaSeems like a match made in heaven! No placebo!MDX 1106 is showing great promise and is a Anti-PD-1.
-
- October 13, 2010 at 10:02 pm
Hi Lisa,
I am currently 6 weeks into the GSK braf trial and even though I realize it is not long term, I am very happy to be completely off my vicodine and not using a cane to walk. I have already done the vaccine, ipi and IL2 with very short term limited results. I guess it all depends on how you look at your situation and what you are willing to go through to manage the beast,
Laurie
-
- October 13, 2010 at 10:02 pm
Hi Lisa,
I am currently 6 weeks into the GSK braf trial and even though I realize it is not long term, I am very happy to be completely off my vicodine and not using a cane to walk. I have already done the vaccine, ipi and IL2 with very short term limited results. I guess it all depends on how you look at your situation and what you are willing to go through to manage the beast,
Laurie
-
- October 14, 2010 at 1:51 am
Hi Lisa,
Is the trial to which you refer NCT01006980? If so, please look at this:
http://www.clinicaltrials.gov/ct2/show/NCT01006980?term=plx4032&recr=Open&rank=1
This trial, being Phase III, incorporates randomization. That should be considered before entering. There are two New York Times articles regarding PLX4032 recently you also might want to look at if you haven't seen them:
http://www.nytimes.com/2010/02/24/health/research/24trial.html
http://www.nytimes.com/2010/09/19/health/research/19trial.html?pagewanted=all
Have you had the v600e testing done yet (i.e. do you meet the key eligibility criteria)? It appears that single path blocking (single agent) may not be as effective as multiple path blocking/inhibiting. Dr Flaherty at Dana Farber/MassGeneral, among others, is looking to combine multiple inhibitors to increase response and durability, but that approach is not yet available in trials yet, to my knowledge. There is some evidence that Ipi will increase the response rate to IL2 significantly, at least one study presented at ASCO, and according to Jim Brietfelter who posts here occasionally.
Best Wishes,
Jim
-
- October 14, 2010 at 11:20 am
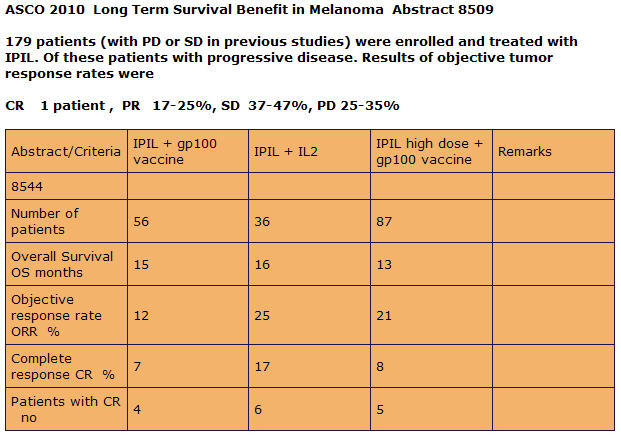
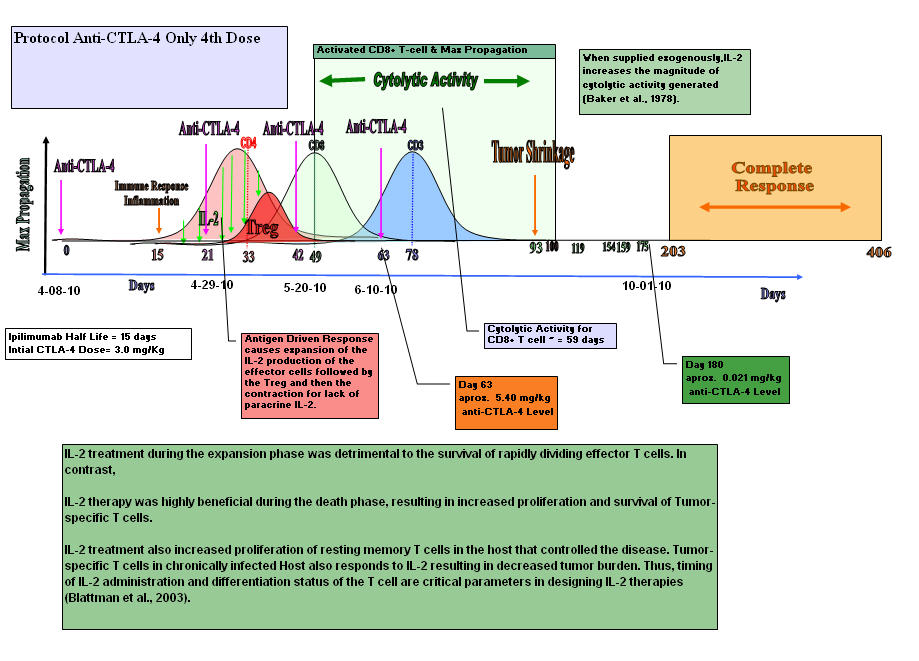
As you can see the combinatorial therapy of Anti-CTLA-4 and Interluekin-2 give the best complete response. I am convince that if they adjust the timing and dose, they will see an even higher response rate in overall survival.
The Making of an Immune Response by Combinatorial Therapy Using Anti-CTLA-4 Blockade and Interleukin-2
The paper that I am about to present to you is a culmination of research that was done base on my own experience as a stage IV Melanoma Patient. I wanted to know why my immune system responded and prove to myself and my doctors that it was not a statistical fluke. I dedicate this paper to all my fellow Melanoma Patients that lost their battle with the Beast, especially Bob Luker who fought so bravely but was unable to obtain Anti-CTLA-4 blockade due to the shortage proclaimed by Bristol Myer Squibb which halted compassionate drug use on 9-12-2008.
Most Melanoma tumors are accepted by the host’s immune system and progress even when they contain potentially antigenic proteins. This may be due to the tumor secreting immunosuppressive Cytokines like, TGF-Beta, IL-10, IL-4 and IL-6. TGF-β inhibits the proliferation and functional differentiation of T lymphocytes. TGF- Beta accelerates the expression of CTLA-4 by stimulated CD4+CD25– T cells. TGF- Beta requires CTLA-4 early after T Cell Activation to induce FoxP3 and generates adaptive CD4+CD25+ (Treg) Regulatory Cells. The tumor cells secrete TGF-Beta.
The Making of an Immune Response by Combinatorial Therapy Using Anti-CTLA-4 Blockade and Interleukin-2
https://www.box.net/shared/n0xcdimy5d
 "><img “It is not the strongest of the species that survives, nor the most intelligent, but the one most responsive to change.”
"><img “It is not the strongest of the species that survives, nor the most intelligent, but the one most responsive to change.”
~Charles Darwin~Take Care,
Jimmy B
-
- October 15, 2010 at 10:09 pm
Hey Jim
Very good information on this. You mentioned you wanted to know why you responded to this tx. My question is just the opposite. I would like to know why my husband did not respond to the Ipi. He was just on Ipi alone but he had finished taking the high dose IL-2 a couple months prior to Ipi without response to that either. I am just confused as to why if it is suppose to take the brakes off does it not work for everyone???? Oh well guess we'll never understand the why's and why not's of Melanoma… Thanks for posting
Linda/Kentucky
-
- October 16, 2010 at 12:11 am
Linda,
My Guess is that the IL-2 therapy grew the the CD4+ T helpers Cells. And a subset of them is the Tregs. These Tregs surpress the immune system. So now your husband has more Tregs in the tumor's microenviroment along with other surpressive cytokines.

Now if you introduce anti-CTLA-4 Blockage it needs to enter the tumor's microenviroment to block all the CTLA-4 receptors on the Tregs. But the CTLA-4 has already bound to the B7 moelcules of the CD8+ T-cells, making the cells inactive. In late stage Melanoma Oncolgists have found an incease of Tregs in the peripheral blood. Your Husband may also be missing the third signal, the "Danger Signal".
(peh-Rperipheral blood IH-feh-rul blud)

Blood circulating throughout the body

Take care
Jimmy B
-
- October 16, 2010 at 12:11 am
Linda,
My Guess is that the IL-2 therapy grew the the CD4+ T helpers Cells. And a subset of them is the Tregs. These Tregs surpress the immune system. So now your husband has more Tregs in the tumor's microenviroment along with other surpressive cytokines.

Now if you introduce anti-CTLA-4 Blockage it needs to enter the tumor's microenviroment to block all the CTLA-4 receptors on the Tregs. But the CTLA-4 has already bound to the B7 moelcules of the CD8+ T-cells, making the cells inactive. In late stage Melanoma Oncolgists have found an incease of Tregs in the peripheral blood. Your Husband may also be missing the third signal, the "Danger Signal".
(peh-Rperipheral blood IH-feh-rul blud)

Blood circulating throughout the body

Take care
Jimmy B
-
- October 15, 2010 at 10:09 pm
Hey Jim
Very good information on this. You mentioned you wanted to know why you responded to this tx. My question is just the opposite. I would like to know why my husband did not respond to the Ipi. He was just on Ipi alone but he had finished taking the high dose IL-2 a couple months prior to Ipi without response to that either. I am just confused as to why if it is suppose to take the brakes off does it not work for everyone???? Oh well guess we'll never understand the why's and why not's of Melanoma… Thanks for posting
Linda/Kentucky
-
- October 14, 2010 at 11:20 am

As you can see the combinatorial therapy of Anti-CTLA-4 and Interluekin-2 give the best complete response. I am convince that if they adjust the timing and dose, they will see an even higher response rate in overall survival.
The Making of an Immune Response by Combinatorial Therapy Using Anti-CTLA-4 Blockade and Interleukin-2
The paper that I am about to present to you is a culmination of research that was done base on my own experience as a stage IV Melanoma Patient. I wanted to know why my immune system responded and prove to myself and my doctors that it was not a statistical fluke. I dedicate this paper to all my fellow Melanoma Patients that lost their battle with the Beast, especially Bob Luker who fought so bravely but was unable to obtain Anti-CTLA-4 blockade due to the shortage proclaimed by Bristol Myer Squibb which halted compassionate drug use on 9-12-2008.
Most Melanoma tumors are accepted by the host’s immune system and progress even when they contain potentially antigenic proteins. This may be due to the tumor secreting immunosuppressive Cytokines like, TGF-Beta, IL-10, IL-4 and IL-6. TGF-β inhibits the proliferation and functional differentiation of T lymphocytes. TGF- Beta accelerates the expression of CTLA-4 by stimulated CD4+CD25– T cells. TGF- Beta requires CTLA-4 early after T Cell Activation to induce FoxP3 and generates adaptive CD4+CD25+ (Treg) Regulatory Cells. The tumor cells secrete TGF-Beta.
The Making of an Immune Response by Combinatorial Therapy Using Anti-CTLA-4 Blockade and Interleukin-2
https://www.box.net/shared/n0xcdimy5d
 "><img “It is not the strongest of the species that survives, nor the most intelligent, but the one most responsive to change.”
"><img “It is not the strongest of the species that survives, nor the most intelligent, but the one most responsive to change.”
~Charles Darwin~Take Care,
Jimmy B
-
- October 14, 2010 at 1:51 am
Hi Lisa,
Is the trial to which you refer NCT01006980? If so, please look at this:
http://www.clinicaltrials.gov/ct2/show/NCT01006980?term=plx4032&recr=Open&rank=1
This trial, being Phase III, incorporates randomization. That should be considered before entering. There are two New York Times articles regarding PLX4032 recently you also might want to look at if you haven't seen them:
http://www.nytimes.com/2010/02/24/health/research/24trial.html
http://www.nytimes.com/2010/09/19/health/research/19trial.html?pagewanted=all
Have you had the v600e testing done yet (i.e. do you meet the key eligibility criteria)? It appears that single path blocking (single agent) may not be as effective as multiple path blocking/inhibiting. Dr Flaherty at Dana Farber/MassGeneral, among others, is looking to combine multiple inhibitors to increase response and durability, but that approach is not yet available in trials yet, to my knowledge. There is some evidence that Ipi will increase the response rate to IL2 significantly, at least one study presented at ASCO, and according to Jim Brietfelter who posts here occasionally.
Best Wishes,
Jim
-
- You must be logged in to reply to this topic.

